Product development
The need for transdermal drug delivery systems is growing every year. In 2019 the global transdermal drug delivery market was valued at 6 billion USD and it is projected to reach 8 billion USD in 2027.
Some of the main advantages driving the development of new transdermal treatments are:
- Avoiding first-pass metabolism, leading to a reduction in applied dosage.
- Ability for a non-invasive and pain-free self-administration by the patient.
- Possibility for improved bioavailability.
- Possibility of maintained, constant, drug concentration over long periods of time
- Elimination of needle injections, which are usually associated with pain, discomfort, risk of infection and thereby results in poor patient compliance.
Thus, transdermal drug delivery is an attractive route across the skin, which enables a systemic effect of drugs, but there are obstacles:
- The major difficulty with transdermal drug delivery is that the outermost layer of the skin, the stratum corneum, constitutes a strong barrier making it difficult for permeants to cross the skin at clinically relevant rates.
- There are currently only ~20 drugs approved for transdermal delivery. Only two new drugs were approved by the FDA between 2000 and 2014.
- No active technology, such as microneedles, electroporation and iontophoresis, has had any commercial success during the last 20 years.
→ Internal development
→ Unique approach
→ Integration of ERCO Pharma’s technology in conventional workflows

Internal development
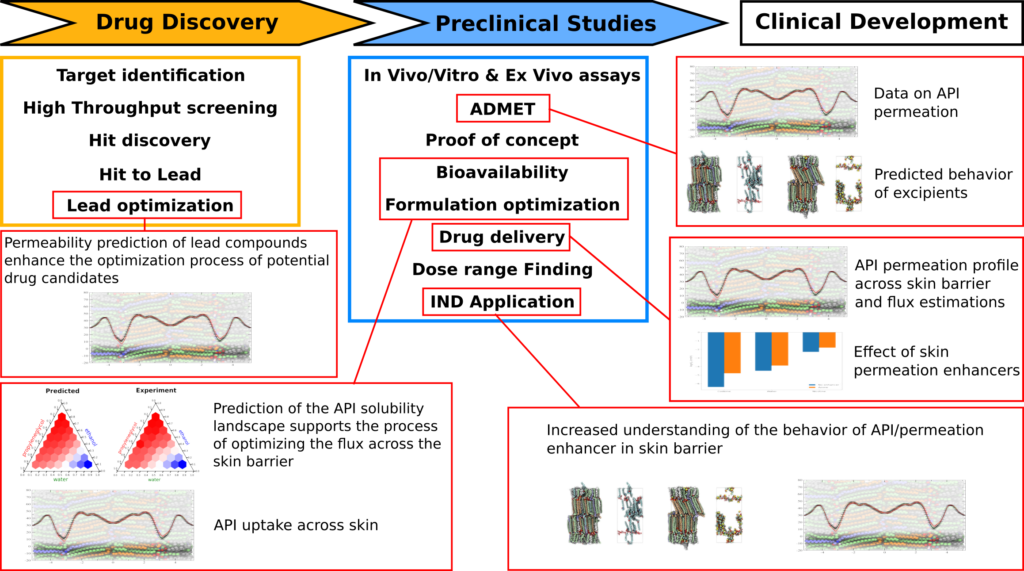
ERCO Pharma currently utilizes its proprietary technology in a number of internal projects primarily aimed at the development of new drug formulations, with the objective to improve the properties of existing drugs on the market.
Utilizing our simulation technology we can predict how, and where, various permeation enhancers interact with the human skin barrier and modify its properties. The collected data enables us to identify combinations of permeation enhancers to be used in formulation development, and the proposed formulations can be tested in silico. From the in silico testing we can then select the most promising candidates for conventional in vitro testing using Franz cells.
Another use of the technology is to study how modifications to an API affect its permeability through skin. In addition to using the technology for increasing the skin permeability it also has the potential to be used to achieve depot effects, by increasing the affinity to specific parts of the skin lipid barrier.


Unique approach
This combined in silico/in vitro approach enables the identification of:
- Which specific regions of the skin barrier that are rate limiting for each API.
- The extent to which each permeation enhancer is responsible for the increased permeation of the API.
- The formulation composition that promotes the highest partitioning of the API into the barrier.
In addition, we are applying our technology to develop novel topical formulations to enable transdermal delivery of drug substances that have previously not been delivered through skin.
The business model for our internal development projects typically involves us taking the projects up to the preparation for Phase I clinical trials. However, we welcome discussions with potential partners as early as possible in the development process.
Integration of ERCO Pharma’s technology in conventional workflows
By integrating molecular modeling in a conventional drug development it is possible to extract additional information from the investigated systems, thus enhancing the decision-making process. High resolution information about the APIs permeation barrier enables us to assess which part of the barrier that needs to be modified by the applied permeation enhancers, and their predicted partition profiles.
An agreement between our molecular modeling data and experimental measurements gives us a higher confidence when deciding whether to take the step towards in vivo testing, compared to the situation when only in vitro data is available.
Since a successful development of a transdermal drug formulation not only requires a solid understanding of the processes involved in the mechanism of action of the API, but also an optimized delivery vehicle across the human skin barrier we believe that the combination of molecular modeling and experimental measurements is paramount in order to design future transdermal products.